|
Published by Oxford University Press for the Infectious Diseases Society of America. Quantitative monitoring of viral load in lower respiratory tract samples helps to evaluate disease progression, especially in cases of low viral load.ĬOVID-19 RT-PCR SARS-CoV-2 ddPCR viral load. Furthermore, the viral loads in the early and progressive stages were significantly higher than that in the recovery stage (46 800 ± 17 272 vs 1252 ± 1027, P <. The viral load of respiratory samples was then compared and the average viral load in sputum (17 429 ± 6920 copies/test) was found to be significantly higher than in throat swabs (2552 ± 1965 copies/test, P <. Four (4/161) negative and 41 (41/67) single-gene positive samples tested by RT-PCR were positive according to ddPCR with viral loads ranging from 11.1 to 123.2 copies/test. In 95 samples that tested positive by both methods, the cycle threshold (Ct) of RT-PCR was highly correlated with the copy number of ddPCR (ORF1ab gene, R2 = 0.83 N gene, R2 = 0.87). Clinical and imaging data were obtained for clinical staging. Nasal swabs, throat swabs, sputum, blood, and urine were collected. The Roche Elecsys® Anti-SARS-CoV-2 S (semi-quantitative) and Anti-SARS-CoV-2 (qualitative) immunoassays detect high affinity antibodies against the spike and nucleocapsid proteins, respectively, to provide high specificitydelivering results you can trust to inform clinical decisions and support patient care. The widely used reverse transcription-polymerase chain reaction (RT-PCR) method has limitations for clinical diagnosis and treatment.Ī total of 323 samples from 76 COVID-19-confirmed patients were analyzed by droplet digital PCR (ddPCR) and RT-PCR based 2 target genes (ORF1ab and N). 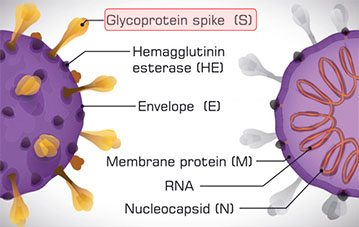
Coronavirus disease 2019 (COVID-19) has become a public health emergency.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
